Ageing promotes microglial accumulation of slow-degrading synaptic proteins
Summary
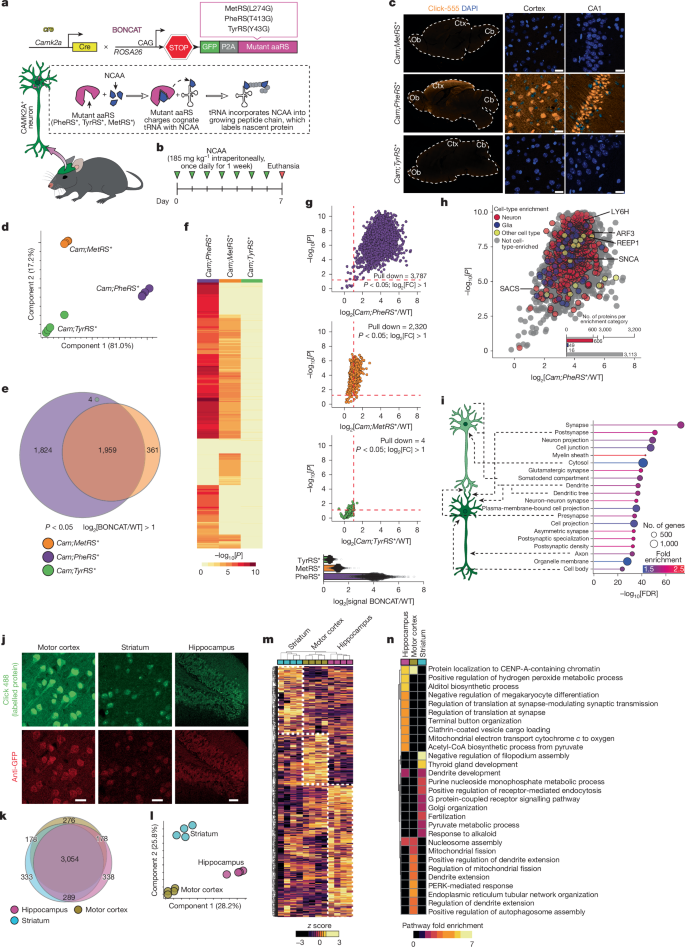
This Nature study introduces new in vivo BONCAT models (PheRS* and TyrRS*, plus comparison with MetRS*) to label nascent neuronal proteins in a cell-specific manner and track their fate across the lifespan. Using these tools (knock-in lines and an AAV:Camk2a-PheRS* vector for aged mice) the authors performed pulse–chase labelling and LC–MS to measure neuronal protein degradation rates, identify proteins that aggregate in aged brains, and map neuron-to-microglia protein transfer.
They find that neuronal protein degradation slows substantially with ageing (on average roughly doubling protein half-lives), with synaptic proteins being particularly prone to slowed turnover and aggregation. By isolating detergent-insoluble aggregates they define a neuronal ‘aggregome’ of >1,700 proteins in aged mice, many of which overlap with human-ageing aggregates and neurodegenerative risk genes. Finally, sorted microglia from aged brains contain large amounts of neuron-derived proteins — heavily enriched for synaptic components — suggesting microglia take up aberrant/old neuronal proteins as a proteostasis mechanism that increases with age.
Key Points
- New BONCAT mouse models (PheRS* performed best for CAMK2A+ neurons) enable robust, cell-specific labelling of nascent neuronal proteomes in vivo.
- Using pulse–chase labelling plus mass spectrometry, neuronal protein degradation slows with age — average half-life roughly doubles from young to aged mice and the deficit appears after middle age.
- Synaptic proteins show disproportionate slowdown in degradation and are over-represented among proteins with the largest age-related half-life increases.
- Isolation of detergent‑insoluble aggregates identified ~1,726 neuronal proteins in aged aggregates (the neuronal ‘‘aggregome’’), with many overlapping human ageing/Alzheimer’s aggregate datasets.
- About half of the proteins with age-reduced degradation are also found in aggregates, implicating aggregation as a contributor (though not the sole cause) of slowed turnover.
- Microglia from aged mice contain many BONCAT-labelled neuronal proteins (mostly synaptic), and aged microglia accumulate far more neuron-derived proteins than young microglia.
- Over 50% of neuron-derived proteins enriched in aged microglia show age-related proteostasis deficits (slower degradation and/or aggregation), suggesting selective transfer of impaired proteins to microglia.
- Regional heterogeneity exists: different brain areas show distinct degradation trajectories and vulnerabilities, which may relate to regional disease susceptibility.
Why should I read this?
Short and blunt: ageing makes certain neuronal proteins — especially at synapses — stick around longer and clump together, and microglia end up hoovering them up. If you care about why synapses fail with age, or how glia contribute to proteostasis and dementia risk, this paper saves you weeks of slogging through mass‑spec papers. It lays out the proteins, the regions and the cell-to-cell handover in clear experimental detail.
Context and relevance
Punchy take: this is a heavyweight, neuron-specific proteostasis map across ageing. The combination of new cell-specific BONCAT labelling, quantitative pulse–chase MS and cell sorting gives direct evidence that ageing compromises neuronal turnover with a synapse-centred signature. That matters because many neurodegenerative risk genes encode synaptic proteins; slowed clearance plus aggregation at synapses offers a mechanistic bridge between normal ageing and disease vulnerability.
Therapeutic implications are immediate: restore neuronal degradation pathways, prevent synaptic aggregation, or modulate microglial clearance capacity could all be viable strategies. But the work also cautions that microglial uptake may be a double-edged sword — clearing damaged neuronal material may protect neurons short-term while overburdening microglia or accelerating synaptic loss long-term. The regional differences highlighted here also argue for targeted approaches rather than one-size-fits-all interventions.