Cholinergic modulation of dopamine release drives effortful behaviour
Article Date: 28 January 2026
Article URL: https://www.nature.com/articles/s41586-025-10046-6
Article Image: 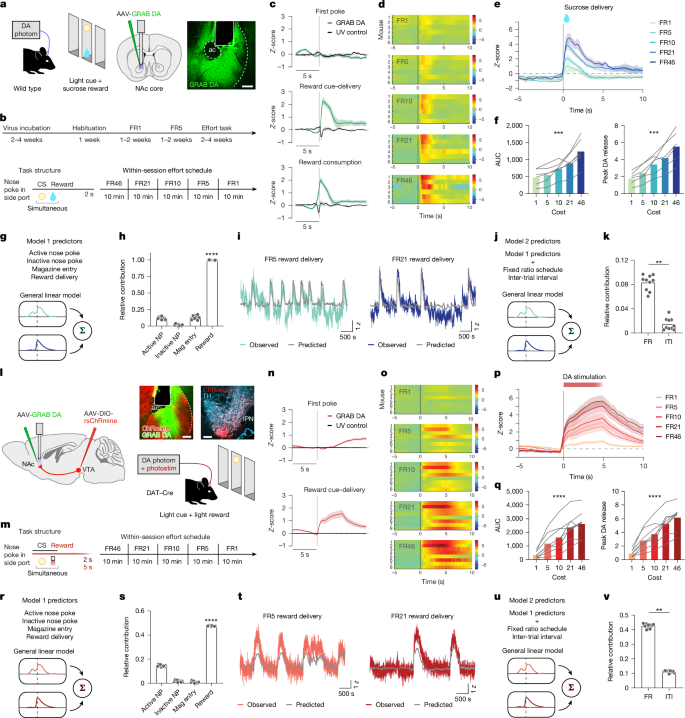
Summary
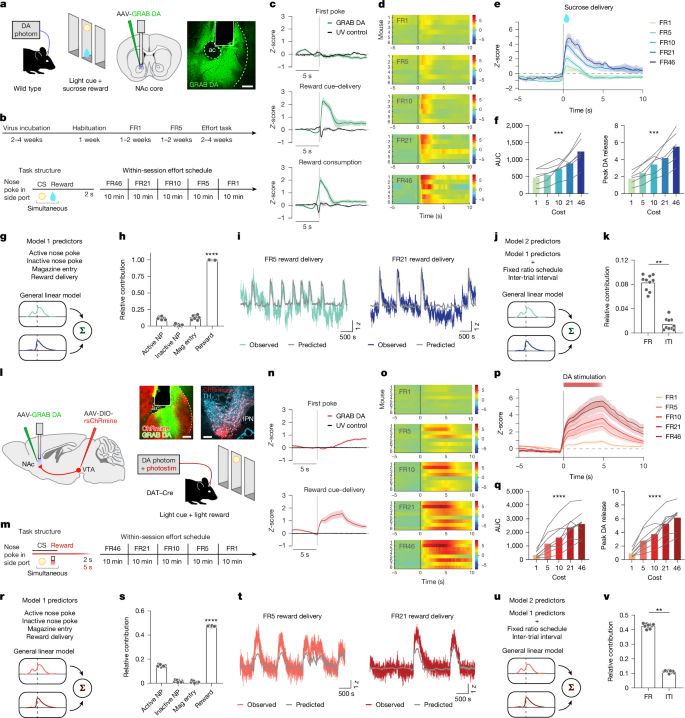
This study shows that dopamine (DA) release in the nucleus accumbens (NAc) scales with the effort expended to obtain a reward, and that this scaling is driven locally at DA axon terminals by acetylcholine (ACh) via nicotinic receptors rather than being strictly controlled by ventral tegmental area (VTA) DA cell bodies. Using GRAB sensors, simultaneous recordings, optogenetic stimulation of DA axons, pharmacology and slice electrophysiology in mice, the authors demonstrate that:
– Reward-evoked DA in the NAc increases with higher fixed-ratio (FR) effort schedules for both sucrose and optogenetic DA stimulation.
– Simultaneous recordings show VTA cell-body activity increases with effort but plateaus earlier than axonal signals and NAc DA release; silencing VTA cell bodies does not abolish the effort-dependent DA increase in the NAc.
– ACh release and cholinergic interneuron activity in the NAc encode effort and precede DA release by ~400 ms.
– Blocking α4/α6-containing nicotinic receptors in the NAc with DHβE, silencing cholinergic interneurons (optogenetically or with tetanus toxin), or otherwise preventing local ACh action selectively blunts the enhanced DA release on high-effort trials and reduces high-effort reward-seeking behaviour.
Key Points
- Dopamine release in the NAc is larger for identical rewards when mice exert more effort (higher FR schedules), for both sucrose and optogenetic DA stimulation.
- VTA DA cell-body activity reflects effort but plateaus sooner and is not required for the full effort-dependent DA increase at terminals.
- NAc acetylcholine (from local cholinergic interneurons) encodes effort and temporally precedes DA release, suggesting a gating role.
- Nicotinic receptors containing α4/α6 subunits are necessary for the effort-related boost in DA; the nicotinic antagonist DHβE abolishes the elevated DA signal at high effort.
- Slice recordings show cholinergic activation can evoke axonal action potentials in DA fibres in the NAc; these responses are blocked by DHβE, confirming a local, receptor-dependent mechanism.
- Disrupting ACh signalling in the NAc reduces animals’ willingness to work for reward at high effort levels, linking the mechanism to motivated behaviour and effort valuation.
Context and relevance
This paper addresses two active debates in systems neuroscience: whether striatal DA release is always a direct readout of midbrain cell-body activity, and how local neuromodulators shape motivational signals. The findings show that local cholinergic signalling can expand the dynamic range of DA release beyond what is driven by VTA firing alone, and that this local ACh→nAChR→DA pathway sustains effortful responding. That matters for anyone studying motivation, reinforcement learning, or addiction — especially since the same nicotinic receptors implicated here are central to nicotine reinforcement. These results suggest new mechanistic angles for understanding why effort can increase subjective value and how drugs of abuse might hijack those processes.
Why should I read this?
Want a quick brain-hack on why slogging for a reward feels more valuable? This paper shows that local acetylcholine in the accumbens turns up dopamine specifically when effort is high — and that blocking those nicotinic receptors makes animals give up sooner. If you care about motivation, reward valuation, or how nicotine affects the brain, this one’s worth a read — it ties the biology directly to behaviour and challenges the idea that midbrain firing tells the whole story.
Author style
Punchy: the study is methodically tight and conceptually important. It doesn’t just add detail — it changes how we should think about the locus of control for effort-dependent motivational signals. For people working on motivation, reinforcement or addiction, the detailed methods and converging manipulations (photometry, optogenetics, pharmacology, slice physiology) are particularly valuable.
Limitations and open questions
- DHβE likely targets DA axon nicotinic receptors but may also affect other inputs (some cortical or thalamic afferents express α4-containing receptors); receptor-specific knockdown on DA axons would be definitive.
- The exact mechanism by which ACh augments optogenetically evoked DA release (recruiting additional axons vs rescuing propagation failures) remains to be clarified.
- Context dependence: ACh–DA interactions may differ across striatal regions, tasks and behavioural states.