Intestinal macrophages modulate synucleinopathy along the gut–brain axis
Summary
This Nature study shows that muscularis externa macrophages (ME-Macs) shape alpha-synuclein (αS) pathology and immune responses along the gut–brain axis in body-first models of Parkinson’s disease (PD). Using 3KL αS transgenic mice and duodenal injections of patient-derived αS fibrils, the authors demonstrate that ME-Macs—but not enteric neurons—engulf misfolded, phosphorylated αS (s129p+), activate endolysosomal programmes and contain seeding-active aggregates. ME-Macs promote local expansion of CD4+ T cells in the myenteric plexus; a fraction of these T cells traffic to the dura mater and brain. Mechanistically, ME-Macs signal to T cells via TGFβ1. Targeted depletion of ME-Macs (anti-CSF1R plus anti-CCR2 injected into the ME) or blocking lymphocyte egress (fingolimod) prevents T cell migration, reduces αS pathology in ENS and CNS, protects dopaminergic neurons and rescues motor deficits in mice.
Key Points
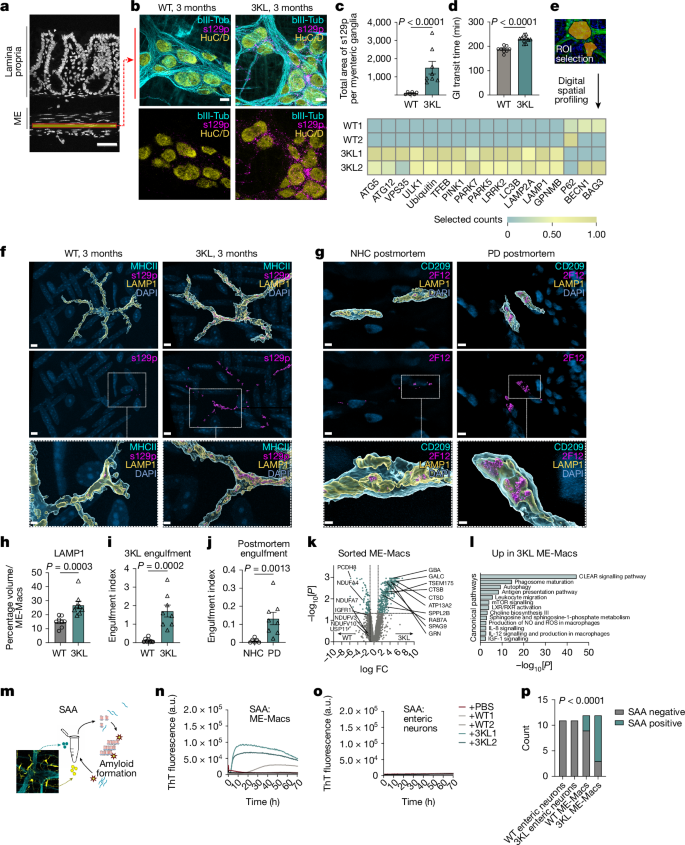
- ME-Macs in the intestinal muscularis externa engulf and accumulate aggregated, phosphorylated αS and show upregulated lysosomal/autophagy pathways.
- αS seeded in the gut (patient-derived PD extracts) propagates to brainstem and substantia nigra and causes dopaminergic neuron loss and motor impairment in mice.
- ME-Macs drive expansion of CD4+ T cells in the myenteric plexus; some intestinal T cells migrate to the dura mater and CNS and share TCR clonotypes with ME T cells.
- TGFβ1 is a top predicted ME-Mac ligand that mediates ME-Mac–T cell crosstalk; deletion of Tgfb1 in self-maintaining macrophages blocks T cell expansion after αS exposure.
- Directed depletion of ME-Macs (anti-CSF1R + anti-CCR2) reduces T cell expansion, lowers s129p+ αS burden in ENS and brainstem, prevents SNpc dopaminergic loss and improves motor behaviour; fingolimod likewise reduces neurodegeneration by blocking T cell egress.
Content Summary
The authors use multiple complementary models and methods: 3KL Thy1-driven αS transgenic mice, duodenal muscularis injections of sarkosyl-insoluble human PD brain extracts, digital spatial profiling, proteomics of sorted ME-Macs, seed-amplification assays (SAA), single-cell RNA-seq, TCR sequencing and targeted immune manipulations.
Key experimental findings: early s129p+ αS accumulates around myenteric ganglia and is enriched in ME-Macs (in both mice and human PD postmortem gut). ME-Macs show elevated LAMP1 and lysosomal proteins and harbour seeding-competent αS in SAA. αS injection into the ME increases gut transit time (constipation-like effect), induces local T cell expansion (CD4 biased), and later leads to αS inclusions in the dorsal motor nucleus of the vagus and selective SNpc dopaminergic loss.
Photoconversion in Vav-H2B-Dendra2 mice and TCR sequencing show that a subset of intestinal T cells migrates to the dura mater and shares clonotypes with ME T cells, implying antigen-driven expansion and trafficking. NicheNet and scRNA-seq identify Tgfb1 from ME-Macs as a prime ligand predicted to affect T cell programmes; macrophage-specific Tgfb1 loss prevents the T cell expansion triggered by PD αS extracts. Finally, local ME depletion of macrophages (anti-CSF1R + anti-CCR2) blocks T cell expansion, reduces αS pathology in gut and brain and rescues motor deficits; pharmacological blockade of lymphocyte egress (fingolimod) also mitigates SNpc degeneration.
Context and Relevance
This paper addresses the ‘body-first’ hypothesis for PD, providing cellular and mechanistic evidence that gut-resident macrophages are early modulators of α-synucleinopathy and of adaptive immune responses that travel to the CNS. It links endolysosomal handling of αS in tissue-resident macrophages to antigen presentation and T cell activation, revealing an immune-driven route for gut-to-brain propagation. The findings intersect several hot topics: peripheral immune contributions to neurodegeneration, the role of tissue-resident macrophages beyond classical clearance, and the gut–brain axis as a therapeutic window.
Why should I read this?
Short answer: if you care about what kicks off Parkinson’s in the gut and how immune cells can drag the problem into the brain, this paper is gold. It’s packed with in vivo models, human tissue comparisons and actionable interventions (macrophage depletion, blocking T cell egress) that actually change pathology and behaviour in mice. Read it if you want a concrete cellular route from gut αS to brain damage — and a few realistic targets to test.