Vagal blood volume receptors compensate for haemorrhage and posture change
Article Date: 28 January 2026
Source URL: https://www.nature.com/articles/s41586-025-10010-4
Article Image: https://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41586-025-10010-4/MediaObjects/41586_2025_10010_Fig1_HTML.png
Summary
This Nature study maps a class of vagal sensory neurons that sense central blood volume and are essential for short-term compensation after posture change (upright tilt) and acute blood loss. Using genetic knockouts, selective ablation, anatomy, electrophysiology and calcium imaging in mice, the authors show that PIEZO2-dependent vagal mechanoreceptors form end-net terminals in the heart, fire with every heartbeat at atrial and ventricular phases, and report changes in central blood volume. Loss of these PIEZO2 neurons or Piezo2 gene function impairs recovery from orthostatic stress and worsens survival in haemorrhage models, demonstrating a protective reflex distinct from arterial baroreceptors.
Key Points
- Researchers identified thoracic vagal mechanoreceptors that detect blood volume changes and are genetically defined by PIEZO2.
- PIEZO2-dependent neurons form end-net endings in the heart (not flower-spray terminals) and fire at two heartbeat-linked phases (atrial and ventricular systole).
- These neurons encode central blood volume: activity falls with blood withdrawal and rises with saline infusion, affecting reflex cardiovascular responses.
- Targeted Piezo2 deletion or ablation of PIEZO2 vagal neurons causes orthostatic hypotension during tilt and impairs compensation after haemorrhage, reducing survival.
- Baroreceptors and PIEZO2 volume receptors collaborate but signal different haemodynamic variables: baroreceptors report pressure, PIEZO2 neurons report central volume.
- Optogenetic stimulation of thoracic PIEZO2 neurons evokes hypotension and mild bradycardia when driven at physiologically relevant frequencies.
Content summary
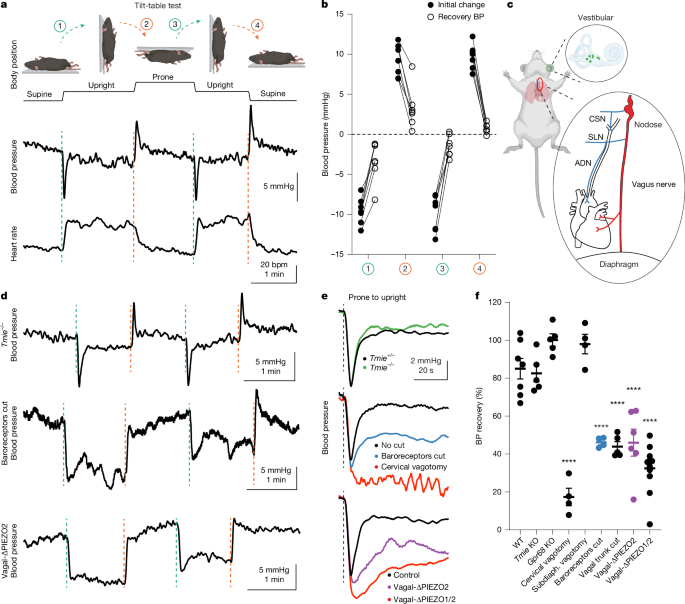
The authors adapted a tilt-table test for mice and combined it with nerve transections, single-cell genetics, AAV tracing, whole-heart imaging, whole-nerve electrophysiology and ganglion calcium imaging. Cervical vagotomy abolished posture compensation; partial denervation suggested a vagal cardiac sensory pathway. Conditional knockout of Piezo2 in Phox2b-expressing cranial sensory neurons (Vagal-ΔPiezo2) impaired gravitational compensation despite an intact arterial baroreflex. Anatomical tracing showed dense PIEZO2+ end-net innervation near atrial vena cava junctions. Electrophysiology of the vagal trunk recorded two heartbeat-synchronised peaks; these were lost with PIEZO2 ablation. Volume manipulations confirmed PIEZO2 neuron activity scales with central blood volume and that their loss leaves mice unable to sustain blood pressure and heart rate during haemorrhage.
The study distinguishes cardiac volume receptors from arterial baroreceptors (which can use PIEZO1 or PIEZO2) and places PIEZO2+ vagal mechanoreceptors as key short-term sensors that help maintain cerebral perfusion on standing and protect against acute blood loss.
Context and relevance
This work clarifies a long-standing physiological question about how the nervous system senses central blood volume and compensates rapidly for orthostatic stress and haemorrhage. It identifies a genetically definable neuronal class (PIEZO2+ vagal afferents) that could be relevant to clinical problems such as neurogenic orthostatic hypotension, syncope and failure to compensate after traumatic bleeding. For researchers, PIEZO2 and the associated end-net terminals offer clear targets for further study; for clinicians, the findings help explain why some patients faint or decompensate after blood loss even when baroreceptor pathways seem intact.
Why should I read this?
Quick version: if you care about how the body keeps blood flowing to the brain when you stand up or after bleeding, this paper is gold. It shows a distinct vagal blood-volume sensor (PIEZO2+) that actually saves mice from fainting and dying in models of tilt and haemorrhage. In short — it’s the missing piece connecting heartbeat-timed nerve firing to real-world protection from orthostatic collapse and trauma.
Author note (style)
Punchy: The authors use rigorous genetic and physiological tools to reveal an essential reflex. This isn’t incremental — it separates pressure-sensing baroreceptors from volume-sensing cardiac mechanoreceptors and links PIEZO2 neurons to survival after blood loss. Read the methods if you want to reproduce the elegant tilt, electrophysiology and imaging approaches.