Mucosal vaccination clears Clostridioides difficile colonization
Article metadata
Article Date: 18 February 2026
Article URL: https://www.nature.com/articles/s41586-026-10138-x
Article image: Figure 1
Summary
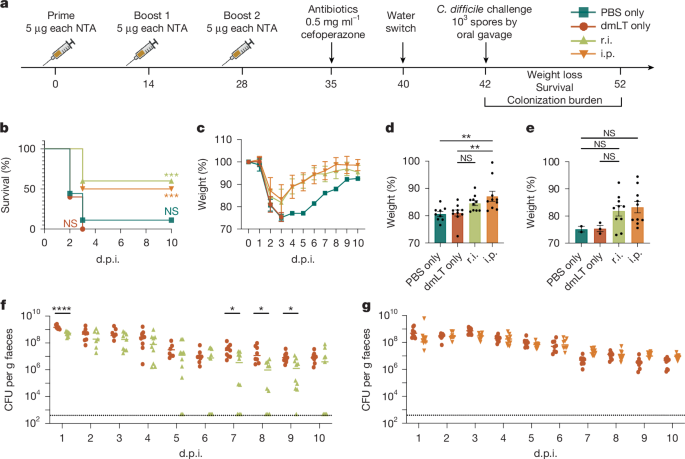
This preclinical study describes a multivalent mucosal vaccine that, when delivered rectally (used here as a mucosal proxy), produces sterilising immunity against Clostridioides difficile (C. difficile) in mice. The formulation combines minimally mutated, natively folded inactivated toxins (TcdA/TcdB) to neutralise toxin-mediated damage, with novel non-toxin antigens (NTAs) targeting vegetative cells and spores (notably CspC, C40 peptidase 2 and a FlgGEK flagellar complex), and uses the double-mutant E. coli heat-labile toxin (dmLT) as a mucosal adjuvant.
The rectal (mucosal) route — but not parenteral (intraperitoneal) vaccination — cleared both vegetative bacteria and spores from the gut, prevented weight loss, epithelial injury and death, and protected against antibiotic-triggered relapse. Mechanistically, clearance correlated with faecal IgG against vegetative surface antigens (which impaired motility) and induction of colonic tissue-resident memory T cells (including CD8+ TRM and CD4+ TRM with TH17-like responses to the spore antigen CspC). The mucosal response was durable (protection and clearance persisted at 60 and 200 days after boost).
Key Points
- Rectal (mucosal) vaccination with a toxin + NTA + dmLT formula produced sterilising immunity in mice, eliminating vegetative C. difficile and spores from faeces and gut tissue.
- Parenteral vaccination (i.p.) protected against severe disease and death but did not clear colonisation — route of delivery was critical for elimination.
- Faecal IgG specific for vegetative antigens (C40 peptidase 2 and FlgGEK) reduced vegetative burden and impaired bacterial motility; systemic IgG alone was insufficient for clearance.
- The spore antigen CspC elicited a mucosal T RM-associated TH17-skewed cellular response that helped target spores and contributed to prevention of relapse.
- Protection and clearance were long-lived: mice challenged 60 and 200 days after boost retained mucosal IgG and TRM responses and cleared infection without relapse.
Context and relevance
C. difficile is a major cause of antibiotic-associated diarrhoea and nosocomial infection with substantial morbidity, mortality and healthcare cost. Previous vaccine efforts focused on injected toxoids or systemic approaches that reduce disease severity but generally failed to clear the gut reservoir — a key limitation because spores transmit faecal–orally and recurrence rates are high. This study demonstrates that combining inactivated, structurally native toxin antigens with surface/spore NTAs and delivering them mucosally (rectally) drives local immune mechanisms — faecal IgG and tissue-resident T cells — that eliminate both vegetative bacteria and spores and prevent relapse. The results matter for vaccine design against enteric pathogens where preventing colonisation and transmission (not just disease) is the goal. The authors suggest a potential human application as an enema-style mucosal vaccine and note public willingness to accept non-standard routes if efficacy is clear.
Why should I read this?
Pretty much: if you care about stopping C. difficile in the gut (not just treating its symptoms), this paper actually shows a way to clear it. It’s preclinical, but the combo of mucosal delivery, surface antigens and natively folded inactivated toxins that together eliminate spores and vegetative cells is a notable step forward — and the immunity lasts months. Read it if you want to know how vaccine route and antigen choice can fix a problem previous vaccines haven’t.
Author note (tone)
Punchy: This isn’t just another toxoid paper — the route and antigen mix change the game. If translation to humans works, it could cut recurrence and transmission, not just hospital stays.