Precancerous niche remodelling dictates nascent tumour persistence
Article metadata
Article Date: 04 March 2026
Article URL: https://www.nature.com/articles/s41586-026-10157-8
Article Image: Figure 1
Author note (style: Punchy)
This is a high-impact mechanistic study. It shows how a fibrotic, fibroblast-driven niche formed at the earliest stages can determine which tiny, nascent tumours survive and which are outcompeted. If you care about cancer initiation, prevention strategies or microenvironment biology, you should read the details — this work points to actionable pathways.
Summary
The Cambridge-led study uses a tobacco-mutagen mouse model (DEN), single-cell RNA sequencing, lineage tracing and 3D heterotypic cultures to show that only a minority of nascent epithelial tumours form a remodelled stromal scaffold — a “precancerous niche” — that promotes their survival and progression. Local PDGFRα-low fibroblasts migrate and clonally expand beneath SOX9-high tumour cells, depositing fibronectin and other extracellular-matrix components. An EGF–SOX9–FN1 signalling axis links stressed epithelial cells and fibroblasts: epithelial EGF ligands (AREG/HBEGF) recruit and activate fibroblasts via EGFR, driving ECM remodelling and fibronectin fibrillogenesis. Blocking EGFR or fibronectin assembly reduces niche formation and early tumour persistence. Key features are conserved in early-stage human oesophageal squamous tumours, underlining clinical relevance.
Key Points
- Most nascent tumours are cleared by competition with neighbouring mutant clones; a minority persist by forming a supportive stromal niche (Niche+ tumours).
- The early niche is built by local PDGFRα-low lamina propria fibroblasts that migrate, clonally expand and deposit fibronectin-rich ECM.
- A distinct epithelial stress state (Tumour 12; marked by high SOX9 and EGF ligand expression) recruits fibroblasts and promotes niche formation through the EGF–SOX9–FN1 axis.
- Ex vivo and in vivo interventions — EGFR inhibition (gefitinib) or blocking fibronectin fibrillogenesis (FUD peptide) — reduce niche formation and nascent tumour survival.
- Single-cell transcriptomes reveal tumour and stromal heterogeneity: niche fibroblasts show a pro-fibrotic, pre-CAF signature before a full CAF phenotype emerges at later invasive stages.
- Human early-stage oesophageal squamous samples recapitulate SOX9/AREG heterogeneity, PDGFRα+ fibroblast proximity and fibronectin deposition, supporting translational relevance.
Content summary
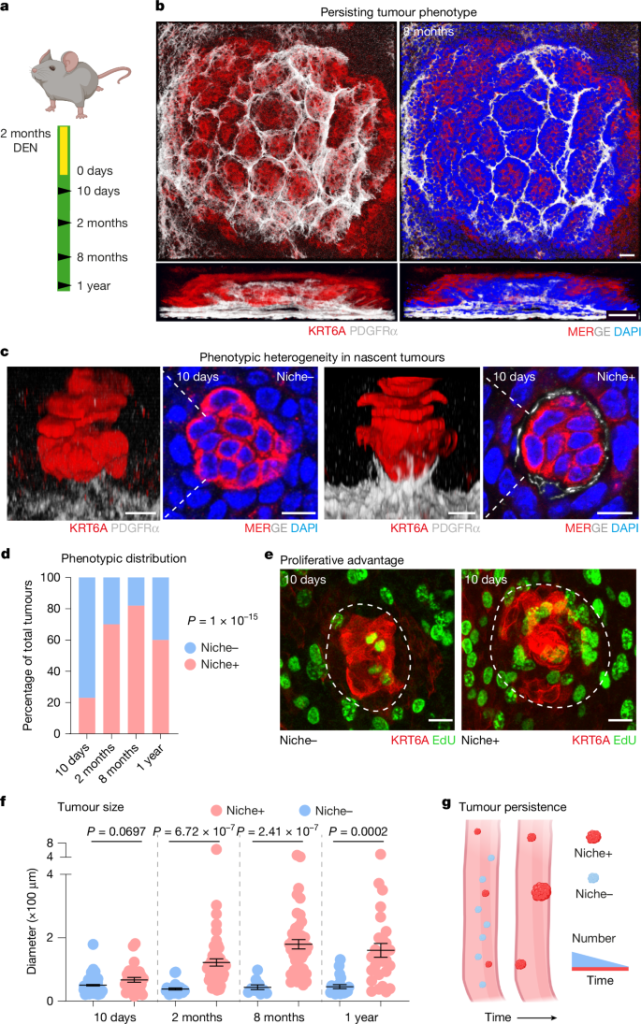
Using a DEN-induced model of upper gastrointestinal tumourigenesis, the authors identify two nascent tumour subtypes: Niche+ (with stromal remodelling) and Niche− (without). Although Niche+ tumours are the minority at formation, they are enriched over time because they are hyperproliferative and more likely to persist and enlarge. High-resolution imaging and lineage tracing show that PDGFRα-low fibroblasts from the lamina propria migrate toward tumour epithelium, clonally expand and form a nest-like scaffold. scRNA-seq places those fibroblasts in a tumour-enriched cluster with upregulated matrisome/wound-healing genes (Fn1, collagens, Loxl2, Mfap5).
Pseudotime and module analyses define a tumour epithelial state (Tumour 12) marked by stress response genes, SOX9 and secreted EGFR ligands (AREG/HBEGF). Cell–cell communication inference predicts strong pro-fibrotic signalling from niche fibroblasts to tumour keratinocytes and EGF signalling from keratinocytes to fibroblasts. Functional assays confirm AREG-driven fibroblast chemotaxis, EGFR-dependent fibroblast compartmentalisation and FN1-driven epithelial proliferation. Pharmacological blockade of EGFR or fibronectin assembly in vivo reduces the number of Niche+ tumours and overall early tumour burden. Human tumour whole-mounts mirror key features observed in the mouse model.
Context and relevance
These data shift part of the focus of early carcinogenesis away from mutation accumulation alone and towards the tissue response to genetic stress. In tissues harbouring many mutant clones, nascent tumours that activate an epithelial stress programme (SOX9/EGF) can co-opt local fibroblasts to build a permissive, fibrotic niche that sustains growth and evades competitive elimination. Targeting niche formation (EGFR signalling or ECM assembly) could therefore be a strategy to prevent precancerous lesions from persisting and progressing. The findings intersect with ongoing interest in tumour microenvironment, cancer prevention and stromal-targeted interventions.
Why should I read this?
Want the short version? This paper gives a neat, experimentally strong answer to why only some tiny tumours survive: they build a fibrotic hideout. It ties a specific epithelial stress state to fibroblast behaviour and ECM remodelling, shows causality with interventions, and validates the signature in human samples — so it’s not just mouse fluff. If you’re into early detection, prevention, or microenvironmental therapies, this saves you hours of digging.