Fibroblastic reticular cells direct the initiation of T cell responses via CD44
Summary
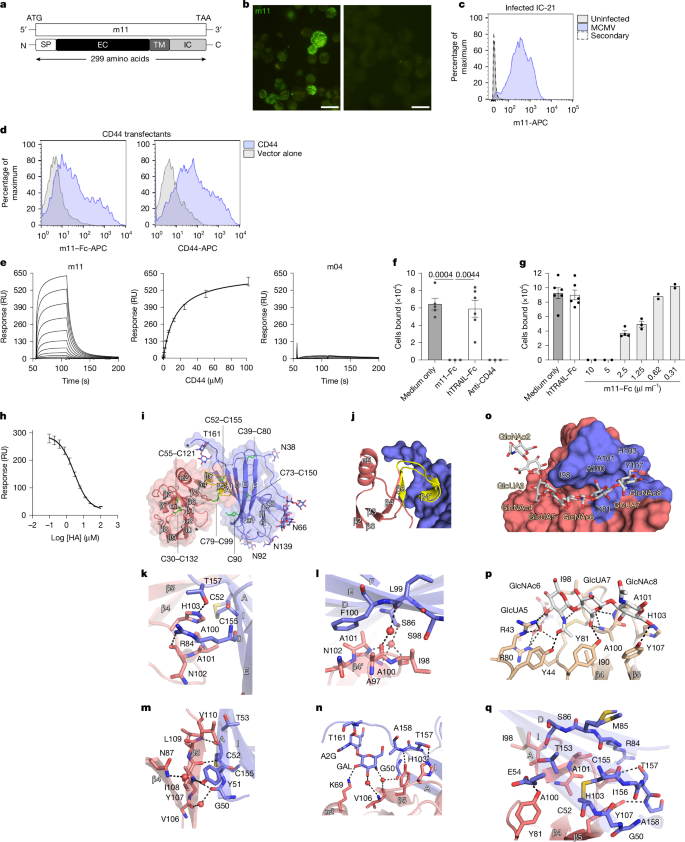
This Nature paper describes how a murine cytomegalovirus (MCMV) protein, m11 (named vCD44BP), binds the cell-surface receptor CD44 and blocks its interaction with hyaluronic acid. The authors show that vCD44BP is expressed on infected stromal cells, notably fibroblastic reticular cells (FRCs), and that this interaction remodels the FRC network, impairs dendritic cell migration into T cell zones and delays antiviral CD8+ T cell priming. Structural biology (crystallography and SPR) defines a direct, competitive interaction between m11 and the hyaluronic-acid binding site of CD44. Genetic and chimera experiments demonstrate that stromal (not haematopoietic) CD44 is the critical target for this immune-evasion mechanism. Soluble vCD44BP also restricts lymph node expansion after adjuvant or infection, and reduces antigen-specific CD8+ T cell responses in influenza and vaccination models.
Key Points
- m11 (vCD44BP) is an MCMV transmembrane protein that binds the CD44 ectodomain with micromolar affinity and occludes hyaluronic-acid binding.
- Crystal structure shows m11 docks onto the CD44 ‘hook’ region, sterically blocking hyaluronic-acid access rather than mimicking it.
- Disruption of m11 (ΔvCD44BP) does not affect in vitro replication but reduces viral loads in vivo from day 6 onwards, a CD8 T cell–dependent phenotype.
- vCD44BP expression in infected FRCs alters CD44 distribution and prevents normal FRC network remodelling, reducing dendritic cell mobility and recruitment into the T cell zone.
- Bone-marrow chimera and CRISPR chimeras localise the critical effect to stromal CD44 rather than haematopoietic CD44.
- Soluble vCD44BP–Fc limits adjuvant-driven lymph node expansion and weakens antigen-specific CD8 responses in vaccination and influenza models — the CD44 pathway has broad relevance beyond CMV.
- Findings expose a new viral immune-evasion strategy and reveal a previously unrecognised role for stromal CD44 in initiating adaptive T cell immunity.
Content summary
The investigators used expression cloning to identify CD44 as the cellular ligand for the MCMV m11 protein and validated binding with cellular assays and surface plasmon resonance. Functional assays showed m11 competes with hyaluronic acid for CD44 binding and blocks hyaluronic-acid-dependent adhesion. The m11–CD44 complex was solved by X-ray crystallography revealing an induced-fit interaction that sterically occludes the hyaluronic-acid site. An MCMV mutant lacking m11 replicated normally in vitro but was controlled more rapidly in vivo via CD8+ T cell mechanisms (IFNγ and perforin dependent).
Mechanistically, vCD44BP acts in cis on infected FRCs: it remains associated with CD44 in stromal cells, prevents CD44 redistribution after infection and alters podoplanin/CD44-associated remodelling of the FRC reticular network. This leads to reduced dendritic cell migration into the white pulp/T cell zone, fewer effective dendritic cell–T cell encounters and an altered balance of effector versus memory precursor CD8+ T cells. Stromal CD44 was shown to be essential for the vCD44BP effect using bone marrow chimeras and CRISPR-modified haematopoietic chimeras. Finally, soluble vCD44BP undermined lymph node expansion and CD8+ responses in vaccination and influenza models, indicating the pathway’s wider immunological importance.
Context and relevance
Why this matters: the paper shifts part of the spotlight in antiviral immunity from classic immune cells to the stromal scaffold that organises immune interactions. It shows viruses can target stromal checkpoints — here CD44 on FRCs — to delay adaptive responses while preserving host survival. That is important for understanding viral persistence and pathogenesis, and for vaccine or immunotherapy strategies that rely on rapid CD8 priming.
Broader implications: CD44 is already of interest in cancer and inflammatory disease. The structural and functional characterisation of a viral CD44 inhibitor suggests new molecular handles for selectively modulating CD44 in disease (either enhancing or dampening immune responses). The FRC–dendritic cell axis is a practical target for improving vaccine-induced T cell priming or, conversely, limiting pathological lymphadenopathy and inflammation.
Author style
Punchy take: this is a high-impact, mechanism-rich study that uncovers viral subversion of a stromal checkpoint essential for kick-starting CD8 immunity. If you care about how adaptive responses are orchestrated in lymphoid tissue (or how pathogens exploit tissue architecture), read it in full — there are structural data, in vivo genetics and translational hooks.
Why should I read this?
Quick take: this one explains how a virus quietly trips up the tissue scaffolding so T cells don’t get the memo fast enough. It’s not just virology trivia — it tells you that stromal CD44 is a real switch for dendritic cell movement and T cell priming, so whether you’re into vaccines, immune‑modulation or viral pathogenesis, it’s worth a skim (and a deeper read if you want the structural and chimera data).