Convergent evolution of scavenger cell development at brain borders
Article Date: 21 January 2026
Article URL: https://www.nature.com/articles/s41586-025-10003-3
Article Image: Figure 1
Summary
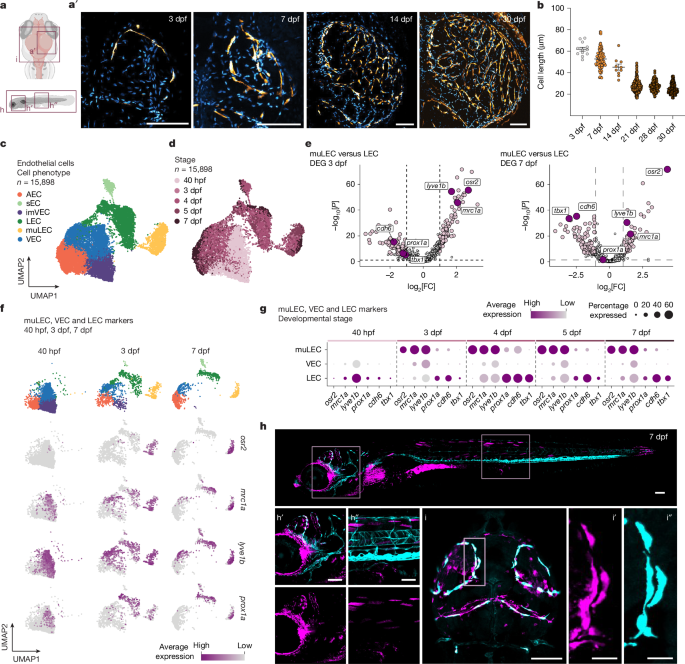
This paper identifies an endothelial lineage in non-mammalian vertebrates — mural lymphatic endothelial cells (muLECs) — that acts as meningeal scavenger cells at brain borders. Using single-cell RNA sequencing, genetic knockouts and cross-species in situ hybridisation, the authors show that the transcription factor odd-skipped related 2 (osr2) is a specific marker and regulator of muLEC differentiation and maintenance in zebrafish. osr2 controls the transition from connected lymphatic endothelial structures to individual, macrophage-like scavenger cells in part by repressing cadherin-6 (cdh6) to reduce cell–cell adhesion.
Comparative analyses show that muLECs are widespread across many vertebrate classes (shark, fish, amphibian, bird), but absent from mammals. Mammals instead possess border-associated macrophages (BAMs), a haematopoietic cell type that performs similar waste-clearing functions. Transcriptomic comparisons demonstrate muLECs and BAMs share scavenger receptor programmes (for example STAB1/2, MRC1, LYVE1) and proteolytic machinery, but the two lineages are developmentally distinct — an example of convergent evolution where equivalent meningeal waste-clearing solutions evolved twice.
The study also shows muLECs respond strongly to injury, contribute to local tissue homeostasis and influence meningeal vascular patterning and innate immune cell numbers. Loss of osr2 impairs muLEC differentiation, reduces scavenging capacity and leads to eventual lineage loss, while altering cdh6 accelerates muLEC isolation and morphology changes consistent with their transition to scavenger cells.
Key Points
- osr2 is identified as a definitive, lineage-specific marker and regulator of zebrafish muLEC development and maintenance.
- osr2 controls the morphological/differentiation switch of muLECs partly by downregulating cdh6; loss of osr2 impairs differentiation and scavenging function.
- muLECs are lymphatic-derived, non-haematopoietic scavenger endothelial cells that are transcriptionally and functionally macrophage-like (high endocytosis, lysosomal and proteolytic gene programmes).
- Cross-species comparison shows muLECs are present in cartilaginous fishes, ray-finned fishes, amphibians and birds but absent from mouse and human meninges.
- Mammals have border-associated macrophages (BAMs) that are haematopoietic and serve analogous scavenging roles; muLECs and BAMs are analogous (functional similarity) but not homologous — a clear case of convergent evolution.
- muLEC loss (genetic or developmental block) alters meningeal vascular patterning and increases local innate immune cell numbers, pointing to physiological roles beyond waste clearance.
- muLECs mount the largest transcriptional response of any meningeal cell type after injury, upregulating endocytosis, proteolysis and lysosomal programmes.
Why should I read this?
Quick take: the brain’s border cleaners evolved twice. If you care about neuroimmunity, lymphatic biology or evolutionary cell biology, this paper explains how different animals solved the same problem — clearing waste from meninges — using different cell origins. It’s a neat mix of single-cell genomics, crisp genetics and cross-species anatomy, and it changes how we think about meningeal immunity across vertebrates. Worth a skim if you want the headline; read the detail if you work on CNS immunity or lymphatics.
Context and relevance
Why this matters: maintenance of meningeal spaces and efficient waste clearance are central to brain health and to diseases such as Alzheimer’s and Parkinson’s where border immunity influences pathology. The finding that muLECs (a vascular-derived lymphatic lineage) perform scavenger functions in many vertebrates, while mammals evolved a haematopoietic BAM lineage to fill the same niche, highlights both evolutionary plasticity and the physiological importance of meningeal cleaning.
Implications:
– For evolutionary biology: demonstrates convergent evolution at the cellular level — functionally similar cell types arising from distinct developmental origins.
– For neuroimmunology: suggests different model organisms may use fundamentally different cellular players to handle meningeal waste and immune signalling; this affects how results from zebrafish or birds translate to mammals.
– For disease research: understanding distinct scavenger programmes (LEC-derived vs BAM-derived) could reveal new targets to modulate meningeal clearance or inflammation in neurological disease.
Overall, the study is both mechanistic (osr2 → muLEC differentiation; cdh6 downregulation) and comparative (multi-species evidence), making it important for researchers working on lymphatics, CNS border immunity and the evolution of immune cell types.