Environmentally driven immune imprinting protects against allergy
Summary
This Nature paper reports that diverse environmental exposures early in life imprint the adaptive immune system and reduce allergic sensitisation in mice. Mice obtained from a pet shop (non‑SPF) carried broad pre-existing type I memory (IgG and IFNγ+ T cells) that made them resistant to experimental allergy and anaphylaxis compared with specific pathogen‑free (SPF) laboratory mice. The protection depended on perinatal environmental exposure: fostering experiments showed a critical early‑life window. The authors show that prior type I or tolerogenic exposures to cross‑reactive orthologues or dietary proteins can both prevent and reverse allergic states by shifting antibody balances (higher allergen‑reactive IgG vs IgE) or by inducing cross‑tolerance. The findings suggest a mechanism linking cumulative environmental antigen experience to lower allergy risk and offer conceptual routes for prevention or therapeutic re‑imprinting.
Key Points
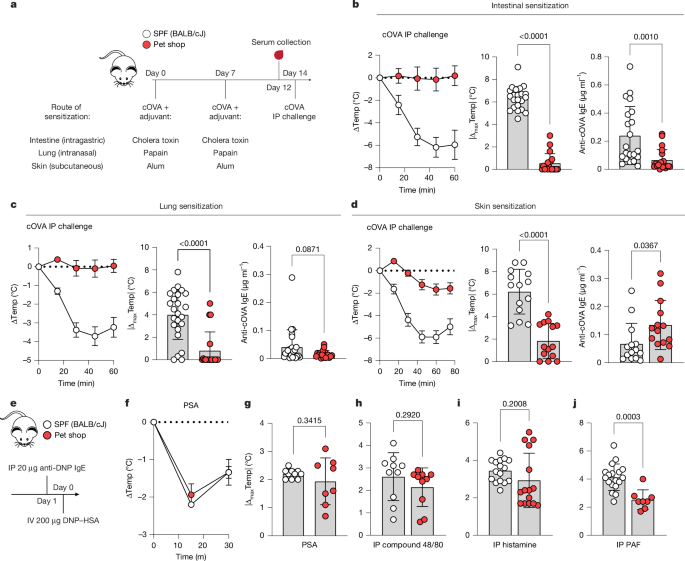
- Non‑SPF (pet shop) mice show strong resistance to experimentally induced allergy and anaphylaxis compared with SPF mice.
- Pet shop mice have pre‑existing cross‑reactive IgG and IFNγ+ memory T cells against model antigens despite no known prior exposure.
- A perinatal window exists: early‑life sensitisation produces type II (allergic) responses, whereas later exposures in an immunostimulatory environment drive mixed type I/type II memory and protection.
- Prior type I immunisation with related orthologues (or infection) shifts responses to IgG-dominant profiles and protects against later allergic sensitisation and even reverses established allergy.
- Oral tolerance and common dietary exposures (for example soy in chow) create cross‑tolerance across related food proteins (pea, peanut), reducing IgE and anaphylaxis.
- Protection correlates with a higher ratio of allergen‑reactive IgG to IgE; passive transfer of IgG can protect allergic mice.
- Adaptive immune imprinting by environmental antigens is proposed as a probabilistic explanation for regional and temporal differences in allergy prevalence.
Content summary
The researchers compared SPF laboratory mice with outbred non‑SPF mice sourced from a pet shop. Pet shop mice carried diverse microbes and pathogens, had elevated baseline immunoglobulins and mounted IgG‑dominant responses after allergen exposure, yielding markedly reduced anaphylaxis. Using epitope display libraries and ELISpot/flow cytometry, they showed pet shop sera and splenocytes contained pre‑existing, cross‑reactive IgG and IFNγ+ memory T cells to model antigens (ovalbumin and KLH) without direct antigen history.
Reciprocal fostering and co‑housing experiments defined a perinatal window: pet shop neonates fostered to SPF dams lost the protective imprint and became allergy‑susceptible as adults, whereas SPF neonates exposed perinatally to pet shop environments gained protection. Further, deliberate type I immunisations with OVA orthologues or Listeria infection in SPF mice imprinted protective memory and increased allergen‑reactive IgG, preventing later allergic sensitisation. Oral tolerance experiments similarly conferred cross‑protection and dietary studies showed that common dietary proteins can induce broad cross‑tolerance to related allergens.
Mechanistically, the work highlights two complementary protective routes: (1) type I‑biased cross‑reactive memory that raises blocking IgG and IFNγ+ T cell responses, and (2) cross‑tolerance that suppresses IgE induction. Both mechanisms imprint across antigenic space and can alter subsequent humoral and cellular responses to related proteins. The authors acknowledge that microbiota, chronic infections, mast cell state and genetics also contribute, and that imprinted protection is probabilistic rather than absolute.
Context and relevance
This study provides experimental support for ideas long suggested by the hygiene and exposure hypotheses: cumulative environmental antigen experience changes adaptive immune setpoints and reduces allergy risk. It links basic immunology (cross‑reactivity, original antigenic sin, IgG vs IgE balance, oral tolerance) to population‑level phenomena — regional variation and the rapid rise in allergic disease. The mouse models recapitulate several features of adult human immunity, making the results relevant for understanding why early‑life environments matter and for exploring interventions that might induce protective imprinting or re‑imprint allergic individuals.
Why should I read this?
Want a straightforward, mechanistic take on why allergies have shot up and what might stop them? This paper nails a neat idea: getting your immune system some varied exposure early (or later, in a controlled way) can train it away from making allergy‑driving IgE. If you care about allergy prevention, vaccination context, or new therapeutic angles that don’t just treat symptoms, it’s worth a quick read — especially the experiments showing you can both prevent and reverse allergic bias in mice.
Author style
Punchy: the authors make a strong case that environment‑driven adaptive immune imprinting is a major, actionable contributor to allergy risk. The work is methodical and broad in scope — from fostering studies to reductionist orthologue immunisations and dietary tolerance tests — and it amplifies the importance of timing (perinatal window) and antigen context. If you follow immunology, allergy research or translational approaches to tolerance induction, the detailed data are well worth diving into.