Robust cytoplasmic partitioning by solving a cytoskeletal instability
Article Date: 28 January 2026
Article URL: https://www.nature.com/articles/s41586-025-10023-z
Article Image: Figure 1
Summary
This Nature paper combines theory, in vitro reconstitution in Xenopus egg extracts and live imaging (zebrafish and Drosophila) to explain how cytoplasm becomes and remains partitioned before membranes form. The authors show that microtubule asters, driven by autocatalytic (branching) nucleation and limited by local inhibition, can self-organise compartments but that this network is intrinsically unstable: faster autocatalytic growth leads to aster “invasion” and coarsening. Two mechanisms prevent collapse: precise cell-cycle timing (synchrony and cycle length that disassembles asters before invasion can develop) and tuning microtubule dynamics so turnover dominates over autocatalysis. Experiments perturbing nucleation and turnover (AurkA beads, Ran(Q69L), MCAK, cycloheximide, morpholinos) confirm the theory and explain why different embryos adopt divergent strategies: large, high-autocatalysis asters in frog and zebrafish enable rapid coverage but require tight cell-cycle control; low-autocatalysis asters in Drosophila are intrinsically stable and fill the cytoplasm gradually over many divisions.
Key Points
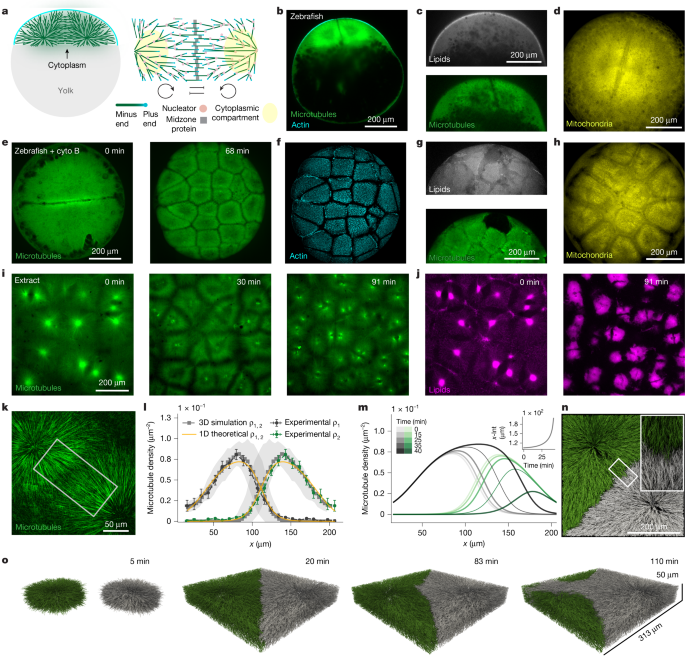
- Microtubule asters partition cytoplasm before membrane ingression in embryos and in cell-free Xenopus extracts.
- A minimal model of two autocatalytic loops with local inhibition reproduces measured microtubule density profiles but predicts a temporal instability (aster invasion and coarsening).
- The instability timescale can be comparable to cell-cycle length; fast, synchronous cycles prevent invasion by disassembling asters before instability develops.
- Stability criterion: compartments are stable when microtubule turnover dominates over autocatalytic nucleation (the paper derives and tests a quantitative inequality linking turnover and autocatalytic rates).
- Altering microtubule nucleation/dynamics changes stability: AurkA beads (lower effective autocatalysis) create stable asters; Ran(Q69L) or high autocatalysis causes instability; MCAK reduces invasion by shortening effective microtubules.
- Cell-cycle arrest (cycloheximide or targeted morpholinos) triggers aster invasion and fusion both in extracts and in zebrafish embryos.
- Zebrafish and frog rely on high autocatalytic growth and precise timing to rapidly cover cytoplasm but risk instability; Drosophila uses low autocatalysis and stable, small asters to partition gradually.
- Combination of theory, 3D agent-based simulations and careful biophysical measurements underpins the conclusions, with code and data publicly available.
Why should I read this?
Short answer: because it sticks physics into a messy biological mystery and actually explains how embryos split their inner stuff before membranes arrive. If you care about cell division, development, or how self-organisation is stabilised in living systems, this paper saves you weeks of digging — neat experiments, clear maths, and a testable stability rule.
Author style
Punchy: the authors don’t just describe a phenomenon — they provide a predictive stability criterion, back it with perturbations and show why different species adopt different strategies. If you’re into mechanisms that explain broad evolutionary and developmental choices, read the full paper.
Content summary
The work documents cytoplasmic compartmentalisation by microtubule asters that occurs independently of cytokinesis in embryos and extracts. The measured microtubule density profiles show exponential-like growth from aster centres (signature of autocatalysis) with local inhibition at interfaces. A 1D continuum model, validated by 3D agent-based simulations, predicts aster invasion: small initial differences lead to one aster progressively consuming its neighbour. The time for invasion depends on initial mass difference and system parameters; if the cell cycle is faster than that timescale and remains synchronous, compartments remain effectively stable. Experimental manipulations confirm the model: arresting the cycle allows coarsening; reducing autocatalytic nucleation or increasing turnover stabilises compartments. Comparing species, zebrafish (and frog) sit in the unstable/high-autocatalysis regime and rely on tight timing, while Drosophila sits in the stable/low-autocatalysis regime and partitions more gradually.
Context and relevance
Why it matters: many embryos and syncytial systems must organise cytoplasm without membranes. This paper provides a mechanistic explanation for how cytoskeletal dynamics and a biological oscillator (the cell cycle) together solve an instability to produce robust spatial patterns. The results link single-molecule microtubule dynamics to organism-scale developmental strategies and suggest evolutionary tuning of nucleation as a lever to change partitioning behaviour. Practical implications include better interpretation of syncytial development, insights for artificial reconstitution of cellular organisation, and a clear quantitative rule to test in other species or perturbations.