The integrated stress response promotes immune evasion through lipocalin 2
Article Date: 18 February 2026
Article URL: https://www.nature.com/articles/s41586-026-10143-0
Article Image: Figure 1
Summary
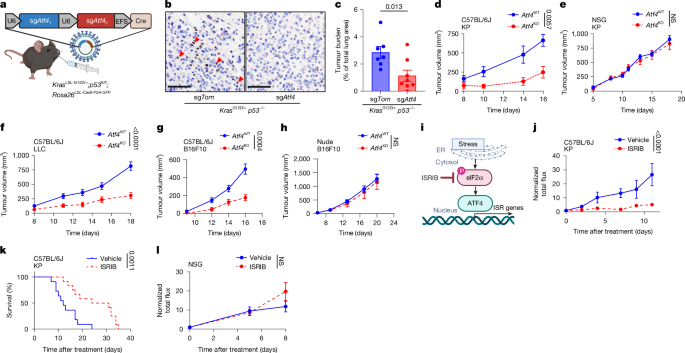
This Nature study shows that the integrated stress response (ISR) — via ATF4 — drives expression and secretion of lipocalin 2 (LCN2) from cancer cells, creating an immunosuppressive tumour microenvironment. Tumour-derived LCN2 binds the macrophage receptor SLC22A17 and reprogrammes interstitial macrophages to a CXCL9-low / IL-6-high state that excludes CD4+ and CD8+ T cells and increases regulatory T cells, enabling immune evasion. Genetic loss of Atf4 or Lcn2, pharmacological ISR inhibition (ISRIB), or neutralising anti-LCN2 antibodies all increase T cell infiltration, slow tumour growth and prolong survival in multiple syngeneic mouse models (lung, melanoma, pancreatic). Importantly, the immunomodulatory function of LCN2 requires secretion but not its iron-binding activity. Human data (tumour microarrays and TCGA/clinical cohorts) link high LCN2 with immune-excluded phenotypes, higher tumour grade, worse survival and poorer response to immune checkpoint inhibitors. The authors also develop anti-LCN2 antibodies that show anti-tumour activity and increase TILs in preclinical models, supporting LCN2 neutralisation as a therapeutic strategy for ICI-refractory solid tumours.
Key Points
- The integrated stress response (ISR) → eIF2α phosphorylation → ATF4 increases LCN2 expression in stressed cancer cells.
- Tumour-derived LCN2 is secreted and signals via macrophage receptor SLC22A17, skewing macrophages to an immunosuppressive state (lower CXCL9, higher IL-6) and increasing T reg accumulation.
- Loss of ATF4 or LCN2, or ISR inhibition with ISRIB, slows tumour growth only in immunocompetent mice — showing the effect is immune-mediated.
- LCN2’s immunosuppressive role requires secretion but not iron–catechol binding; mutants that block secretion lose function.
- Anti-LCN2 antibodies (mouse- and human-specific clones) increase CD4+/CD8+ TILs, reduce tumour burden and, combined with anti-PD1, further extend survival in mouse models.
- Human tumour analyses (LUAD, PDAC, TCGA pan-cancer) show high LCN2 correlates with immune-excluded phenotype, greater macrophage density, fewer intratumoural T cells and poorer outcomes, including reduced benefit from immunotherapy.
Content summary
The authors used genetically engineered mouse models, syngeneic transplant models and pooled CRISPR screening of ATF4 targets to identify Lcn2 as a top ATF4-driven mediator of immune evasion. ATF4-deficient tumours grew poorly in immunocompetent mice but not in immunodeficient hosts; ISRIB phenocopied genetic ATF4 loss. LCN2 knockout reduced tumour growth and increased CD8+/CD4+ T cells while decreasing FOXP3+ T regs and interstitial macrophage subsets. ExCITE-seq and functional assays showed tumour-derived LCN2 reprogrammes myeloid cells and suppresses CXCL9 production, limiting T cell recruitment. Human tissue microarrays and AI-based spatial analysis linked local LCN2 expression to T cell exclusion and worse prognosis. Finally, synthetic neutralising antibodies against LCN2 reduced tumour growth, promoted TIL infiltration and improved survival in preclinical models, and combining anti-LCN2 with PD-1 blockade gave additive survival benefit in mice.
Context and relevance
Why this matters: immune checkpoint inhibitors have revolutionised oncology, but many solid tumours remain resistant. This paper reveals a stress-response-driven, tumour-cell-intrinsic mechanism that actively excludes adaptive immunity via a secreted factor (LCN2). Blocking this axis — genetically, pharmacologically (ISRIB) or with antibodies — restores T cell infiltration and slows growth in otherwise immunologically cold, ICI-refractory models. For translational researchers, oncologists and biotech developers, LCN2 is a compelling, druggable node connecting tumour stress responses to macrophage-mediated immune suppression. The finding also gives a mechanistic explanation for why metabolic or hypoxic tumour stress might blunt immunotherapy efficacy and suggests combination strategies (LCN2 neutralisation + ICI) worth pursuing clinically.
Author style
Punchy: this is a significant translational advance — they link a conserved stress pathway (ATF4) to a secreted immune-suppressive effector (LCN2), show causality across models and back it up with human data and an actionable antibody. If you work on immunotherapy resistance, myeloid biology or antibody development, read the full paper.
Why should I read this?
Quick and dirty: tumour cells under metabolic or ER stress switch on ATF4 and secrete LCN2, which mugs macrophages and keeps T cells out. The team not only proves it in multiple mouse models and patient samples but also shows you can target LCN2 with antibodies to re-open tumours to immune attack. If you care about why some tumours are ‘cold’ or how to make immunotherapy work better, this paper saves you time — and points to a concrete therapeutic route.