Climbing fibres recruit disinhibition to enhance Purkinje cell calcium signals
Summary
This study shows that cerebellar climbing fibres (CFs) preferentially engage a specific molecular-layer interneuron (MLI) subtype (MLI2) through glutamate spillover. Activated MLI2s inhibit MLI1s, producing disinhibition of Purkinje cells (PCs). The disinhibitory pathway (CF→MLI2→MLI1→PC) amplifies CF-evoked dendritic calcium transients in PCs, especially when multiple CFs fire synchronously, creating conditions that favour long-term depression (LTD) at granule cell (GrC)→PC synapses and supporting cerebellar learning.
Key Points
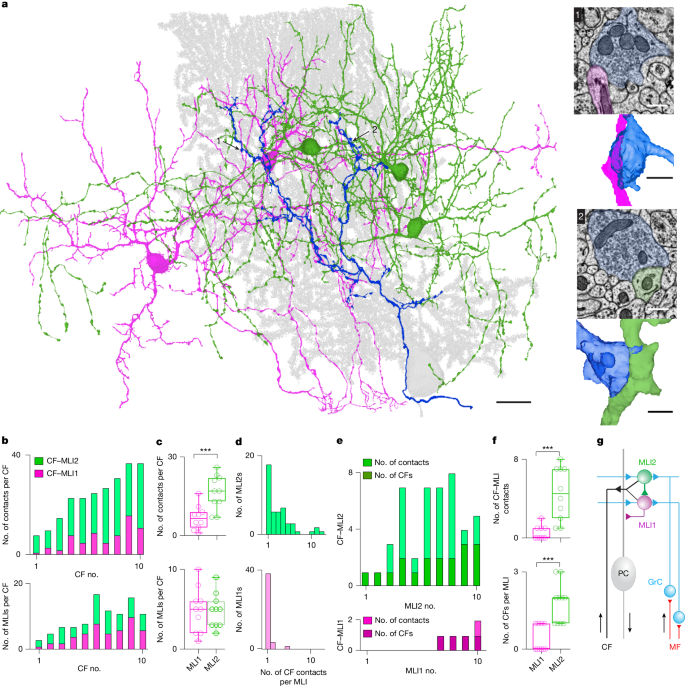
- Serial EM reconstructions reveal CF boutons make more and larger contacts onto MLI2s than MLI1s; MLI2 contacts are closer to GrC PSDs, promoting spillover.
- Electrophysiology shows CF→MLI2 responses are large, slow, all-or-none and have prominent NMDA and AMPA components; CF→MLI1 spillover is weaker and largely AMPA-mediated.
- MLI2s inhibit MLI1s; because MLI1s normally suppress PCs, CF-driven MLI2 activation produces net disinhibition of PCs.
- In vivo Neuropixels recordings and simulations indicate spontaneous CF activity strongly excites MLI2s, causes mixed short-latency effects in MLI1s, and leads to delayed increases in neighbouring PC simple-spike firing consistent with disinhibition.
- Sensory-evoked CF synchrony (multiple nearby CFs firing together) greatly enhances PC dendritic calcium transients — both by driving multiple CF spikes and by recruiting the disinhibitory circuit — thereby creating ideal conditions for LTD and learning.
Content summary
The authors combined serial electron microscopy, acute slice electrophysiology, in vivo high-density recordings, two-photon calcium imaging and large-scale network simulations. EM reconstructions from lobule V show each CF contacts roughly equal numbers of MLI1s and MLI2s but makes many more contact sites per MLI2; contact areas onto MLI2s are larger and sit nearer multiple GrC PSDs. These anatomical differences predict stronger CF-driven spillover onto MLI2s.
Slice recordings confirm robust CF-evoked spillover currents in MLI2s with clear NMDA contributions, whereas CF→MLI1 spillover is weaker and AMPA-dominant. The presence of Grin2b (NR2B) in MLI2s explains the NMDA sensitivity and links MLI2 activation to plasticity mechanisms. Reconstructions and simulations show that MLI2s, once activated, inhibit many MLI1s; given MLI1s preferentially inhibit PCs, this yields net disinhibition of local PCs.
In behaving mice, spontaneous CF events (proxied by Purkinje complex spikes) evoke rapid increases in MLI2 firing, mixed fast effects on MLI1s, and a biphasic effect on neighbouring PCs (very fast suppression followed by slower increased firing). Two-photon imaging demonstrates that sensory-evoked CF synchrony or multiple CF spikes produce substantially larger PC dendritic calcium signals than isolated CF events. Simulations recapitulate these dynamics and show that CF synchrony biases the network towards the disinhibitory pathway, enhancing calcium transients and thus the likelihood of LTD at GrC→PC synapses.
Context and relevance
This work resolves an apparent paradox: CFs both excite Purkinje cells (driving calcium spikes needed for plasticity) and activate interneurons that can suppress those same calcium signals. The discovery that CFs preferentially recruit MLI2-mediated disinhibition explains how CFs can simultaneously trigger strong dendritic calcium signals and relieve inhibitory gating for a brief window — a mechanism readily aligned with cerebellar associative learning theories. The paper ties precise microcircuit anatomy to functional outcomes (calcium signalling and plasticity), showing how synchrony in olivary/CF activity can control the gating of learning at a fine spatial scale (parasagittal microzones).
Why should I read this
Want the short version — CFs don’t just whack Purkinje cells: they cleverly recruit a disinhibitory microcircuit that amplifies the calcium signal exactly when learning needs it. If you care about how microcircuit motifs gate plasticity, or how timing and synchrony shape learning signals in the cerebellum, this paper packs anatomy, physiology, imaging and modelling into a single mechanistic story. Saves you hours of digging; gives you a crisp new circuit to think with.
Author style
Punchy: the authors nail both the anatomical substrate and the functional consequence. They demonstrate a clear circuit motif — CF→MLI2→MLI1→PC — that explains how climbing-fibre signals are selectively amplified by disinhibition. If you work on cerebellar plasticity, interneuron diversity, or disinhibitory motifs in neural circuits, the details here are directly relevant and worth diving into.
Source & Article meta
Article Date: 18 March 2026
Article URL: https://www.nature.com/articles/s41586-026-10220-4
Source: Nature — Climbing fibres recruit disinhibition to enhance Purkinje cell calcium signals

